On July 15 2025, to mark the successful secondary listing of China Medical System Holdings Limited (“CMS” or the “Group”) on the Main Board of the Singapore Exchange (“SGX”), SGX and CMS co-hosted the “Singapore and Emerging Markets Pharmaceutical Industry Growth Forum & CMS SGX Secondary Listing Appreciation Dinner”. Held in a grand fashion, the event was held at the Group’s CDMO manufacturing facility, PharmaGend, which is located in Tuas, Singapore.
The event brought together about 150 representatives from local government agencies, multinational pharmaceutical companies, innovative biotech companies, leading investment institutions, and the KOLs in the pharmaceutical industry. Through a series of insightful keynote speeches and panel discussions, guests engaged in in-depth exchanges and shared ideas on various topics, such as the pharmaceutical industry’s development in Singapore and emerging markets across the Asia-Pacific region, the breakthroughs and overseas expansion of Chinese innovative drugs, the globalization strategies, commercialization pathways, as well as ecosystem collaboration of innovative pharmaceutical companies.
The forum began with opening remarks by Ms. Caihan Chia, Head of Greater China Capital Markets and Chief Representative of Beijing Representative Office at SGX, and Ms. Louise Ho, Assistant Vice President of Healthcare Division and China Desk at the Singapore Economic Development Board. These were followed by keynote addresses from Mr. Siang Sheng Foo, Head of Investment Banking at Singapore CGS International Securities, Mr. Shriharsha Sarkar, Partner for Asia Healthcare at L.E.K. Consulting, and Ms. Kah Yean Neo, Senior Director at Singapore’s Agency for Science, Technology and Research (A*STAR).
Ms. Caihan Chia stated that in recent years, SGX has become increasingly attractive to Chinese enterprises through policy refinements, including tax incentives, capital support from the secondary market, and streamlined regulatory procedures. The successful listing of CMS showcases the growing interest among Chinese companies in the Singapore market. As one of leading healthcare companies, CMS’s listing highlights the growing demand for medical innovation and medical service accessibility across Asia. With CMS seeking to expand its business in Southeast Asia, its listing on SGX will serve as a strategic springboard to connect with international investors and further reinforce Singapore’s role as a vital capital hub.
Emerging Markets: A New Growth Engine for the Global Pharmaceutical Industry
Emerging markets, such as Southeast Asia and the Middle East, are becoming new growth drivers for the global pharmaceutical industry. A combination of factors, including large populations, early signs of ageing, the rise of the middle class, growing health awareness, and the increasing burden of chronic diseases, is driving higher demand for medicines and improved accessibility. According to IQVIA, by 2028, the combined pharmaceutical market size of four major emerging regions – Asia-Pacific, India, Africa & the Middle East, and Latin America – is expected to reach USD 336 – 384 billion, comparable to the USD 410 billion market size projected for Western Europe.
Singapore possesses geographical and institutional advantages for accessing Southeast Asia, the Middle East, and other emerging markets. With its robust financial system, open and inclusive policy environment, and thriving pharmaceutical industry, Singapore is increasingly becoming a global hub for capital and innovation. It has also become the preferred location for regional headquarters for many Chinese enterprises expanding into Southeast Asia.
Seizing Opportunity: Strategic Pathways for Chinese Innovative Pharma to Expand into Emerging Markets
In Southeast Asia’s six major economies (SEA6), limited healthcare coverage means that out-of-pocket payments constitute the primary source of drug expenditure. While generics dominate, branded originator drugs continue to hold significant market share in private hospitals, retail pharmacies, and clinics. Patient demand for biologics and biosimilars continues to grow.
In terms of commercialization models, traditional distribution model, which relies on third-party logistics (3PL), is gradually giving way to models with stronger commercial capabilities and strategic licensing partnerships. To achieve sustainable success in Southeast Asia, pharmaceutical companies must build competitive product portfolios, leverage experienced local sales teams, and consider establishing localized manufacturing capabilities, widely seen as key strategic advantages.
The CMS’s Approach: Building Dual Hubs in China and Singapore to Drive End-to-End Innovation
With over 30 years of experience in the Chinese market, CMS has accumulated a differentiated product portfolio and mature commercialization capabilities. Today, the Group is expanding its strategic vision across the Asia-Pacific region, using China as a foundation and Singapore as its regional hub. Through an end-to-end value chain of “R&D–manufacturing–commercialization–investment”, CMS is driving innovation to deliver high-quality pharmaceutical products and services to patients worldwide.
Mr. Lam Kong, Chairman, Chief Executive and President of CMS, delivered a keynote speech titled “New CMS, New Ascent: Three Strategies to Drive the Second Growth Curve.” He shared that since launching its “New CMS” transformation strategy in 2018, the Group has propelled growth through three engines — product innovation, commercial transformation, and international expansion. This has enabled the Group’s transition from “China’s largest CSO” to “a pharmaceutical company in transformation,” and finally, to “an end-to-end innovative pharmaceutical enterprise”, with a sustainable second growth curve.
In product innovation, driven by a three-dimensional approach of “Licensing, Strategic partnerships, and in-house R&D”, the Group has built a pipeline of nearly 40 FIC/BIC innovative drugs, five of which have been approved in China and are in large-scale clinical use. In the area of commercialization, CMS remains focused on cardio-cerebrovascular, gastroenterology, ophthalmology, and skin health specialties, while enhancing anti-cyclical resilience through a diversified ecosystem of “New retail, E-commerce, and Consumer healthcare”. Its skin health subsidiary, Dermavon, has become a niche market leader in China and is now progressing toward a spin-off for an independent listing on the Hong Kong Stock Exchange. In the area of globalization, CMS is creating a dual-track model centred in China and Singapore, using a strategy of “bringing in” to accelerate overseas product launches in China, and a strategy of “moving outward” to establish an end-to-end presence in emerging markets. The successful listing on SGX will enhance its regional synergy and close the loop in the “R&D – Manufacturing – Commercialization – Investment” global value chain, unlocking growth from emerging markets and creating a multi-regional growth framework.
CMS formally launched its industrial globalization strategy in 2022. At this event, CMS’s international business clusters made their debut, showcasing its forward-looking, full industry chain layout and leadership in setting a new paradigm for Chinese pharmaceutical companies expanding overseas.
PharmaGend
Established in 2023, PharmaGend aims to become Southeast Asia’s largest and most reliable CMO/CDMO. It has a site spanning 30,000 square meters and is capable of manufacturing dosage forms such as tablets and capsules, which has been certified by the FDA and HSA, demonstrating its high-standard pharmaceutical manufacturing capabilities for global export. It has future plans to expand production lines for injections, ointments, and nasal sprays.
Rxilient
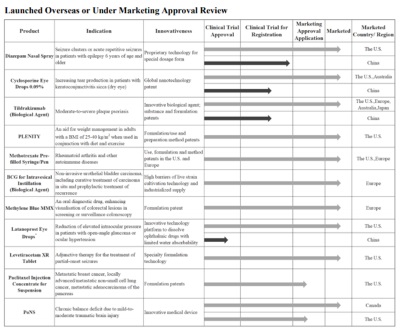
Established in 2021, Rxilient operates by a professional and experienced localized team, and has fully established BD, registration, marketing, andcommercialization capabilities. Leveraging its unique local expertise and advantages, Rxilient can bring innovative drugs to emerging markets led by Southeast Asia and the Middle East. It has submitted marketing applications for nearly 20 drugs and medical devices across Southeast Asia, the Middle East, and regions such as Hong Kong, Macao, and Taiwan, covering the therapeutic areas of dermatology, ophthalmology, oncology, autoimmune, and central nervous system. As more drugs receive regulatory approval in these countries, Rxilient anticipates sustained and significant revenue growth.
CMS R&D
Established in Singapore in 2024, CMS R&D has been working on more than 10 early-stage innovative drug projects. Leveraging China’s mature early-stage R&D and clinical resources, it aims to synchronize China speed with global standards to advance more Chinese innovative drugs toward globalization.
HiGend
Established in 2025, HiGend is a global early-stage bio-pharma incubation platform which uses a “hub-and-spoke” model, integrating China’s innovation capabilities to accelerate global R&D and commercialization.
Subsequently, three panel discussions were held in succession, which facilitated an in-depth exchange between industry and capital, driving the forum to its climax. Distinguished guests from various parties freely shared insights on the continuous development and diversification of the pharmaceutical ecosystem, and jointly explored the future of pharmaceutical expansion into emerging markets.
Panel Discussion 1 – Challenges and Breakthroughs: The Enduring Power of Organizational and Strategic Long-Term Vision
The emerging markets of today share numerous similarities with China’s pharmaceutical landscape twenty years ago, which are currently experiencing a critical period of accelerated demand release for pharmaceuticals, constituting medium- to long-term structural opportunities. Undoubtedly, these markets are diverse and complex — each country has its own unique characteristics in terms of drug regulation, healthcare insurance mechanisms, and market acceptance. However, CMS’s core strength lies in its systematic commercialization capabilities, which it is now extending to emerging markets. CMS’s senior management team, alongside its business partners, jointly reviewed and discussed the key factors contributing to its commercial excellence, as well as the pathways driving the Group’s second growth curve.
The first panel discussion was moderated by Mr. Brian Yang, Vice President for Business Development at Rxilient. Participants included Mr. Karl Luschmann, Managing Director of Pharma Stulln GmbH, and Ms. Linlang Wang (formerly the first product manager of the Augentropfen Stulln Mono Eye Drops (“Stulln”) in China), General Manager of CMS’s ophthalmology business, CMS Vision, among others. Collectively, they reviewed the core strategies behind the rapid, year-on-year growth of Stulln in the Chinese market- a focus on clinical value and continuous innovation in commercialization models. CMS adhered to prioritizing clinical efficacy, amassing substantial evidence to demonstrate the clinical value of Stulln in treating asthenopia, and leveraging medical advancements to drive product commercialization. Meanwhile, CMS also continuously revamped its commercialization model by establishing a full-channel retail system that integrates in-hospital and out-of-hospital sales, developing an “online + offline” omnichannel marketing system, and adopting a diversified product portfolio strategy in consumer attributes. These concerted efforts facilitated the sustained, rapid growth and wide recognition of Stulln within the Chinese market.
Mr. Victor Yin, Country Manager of Incyte Bioscience China, Mr. Huang Anjun, CEO of Dermavon (CMS’s skin health business), and Mr. Lawrence He, CEO of Rxilient, jointly retraced the entire journey of launching ruxolitinib cream — a blockbuster prescription drug with consumer attributes. From the signing of the collaboration agreement and pilot launch in Hainan Boao Lecheng pilot zone, to marketing approvals in Macao, followed by Hong Kong, introduction into designated hospitals in the Greater Bay Area in China, and the NDA has been submitted in China, Singapore, and other countries or regions. Leveraging mature clinical development experience and capabilities, proven commercialization competence, a compliant operational system, and efficient execution, the group earned high recognition and trust from Mr. Victor Yin.
Together, the parties have helped bring new hope to vitiligo patients in both China and Southeast Asia simultaneously.
These successful commercialization experiences not only provide valuable business model references for international pharmaceutical companies entering the Chinese market, but also offer significant insights for Chinese pharmaceutical companies looking to expand into emerging markets overseas.
Panel Discussion 2 – Breaking Through: Diverse Explorations for Chinese Innovative Pharmaceutical Companies to Expand into Emerging Markets
Over the past three years, Chinese pharmaceutical companies have secured over USD 10 billion in upfront payments through license-out deals. However, the majority of these transactions remain concentrated in mature markets led by Europe and the United States. Looking ahead, the next engine of growth may shift toward emerging markets such as Southeast Asia, the Middle East, and Latin America — regions with a combined population of approximately 1.8 billion and per capita healthcare spending is merely one-fifth that of Western markets. While pharmaceutical demand is accelerating in these areas, challenges persist, including limited payment capacity and significant differences in regulatory systems. Whether Chinese pharmaceutical companies can effectively replicate and localize their domestic development and innovation models in these blue ocean markets, will determine the scale and sustainability of their second growth curve.
The second panel discussion, moderated by Mr. Brian Yang, featured esteemed representatives from leading Chinese innovative pharmaceutical companies that are closely collaborating with CMS, including Tibet Nordicon Pharma, NeuroDawn Pharmaceutical (Ningdan Pharmaceutical), Mabgeek Biotech, and Jingze BioPharmaceutical. Using examples such as XinHuoSu (for acute decompensated heart failure), Y-3 for Injection (under development for stroke treatment), ABP-671 (under development for gout) etc., participants held in-depth discussions on topics, including “What constitutes truly clinically valuable innovation” and “How to implement commercialization pathways within emerging markets”
True innovation value stems from professionalism and dedicated focus. It requires researchers to remain committed to a specific field over the long term, and to validate new targets and drug structures through reverse translational research, thereby identifying their potential clinical value. Since China officially joined the International Council for Harmonization of Technical Requirements for Pharmaceuticals for Human Use (ICH) in 2017 and became a member of its steering committee, the quality of Chinese innovative pharmaceutical products has improved rapidly and has gradually gained global recognition. In addition to entering mature markets led by Europe and the U.S., Chinese innovators are increasingly turning their attention to emerging blue ocean markets such as Southeast Asia, actively exploring parallel regulatory filings in both emerging and mature markets. For innovative pharmaceutical companies, globalization strategies should be integrated into the early stages of pipeline development cycles and macro-level strategy. It is also crucial to seek out a business partner that possesses the entire value-chain capabilities — including experienced local clinical registration teams, manufacturing capacity, and commercialization operations—in order to drive efficient product launches and expand access to high-quality medical solutions across a broader range of geographies.
Panel Discussion 3 – Setting Sail: Pharmaceutical Expansion into Emerging Markets
The third panel discussion was moderated by Mr. Frank Hong, Managing Director of Legend Capital, engaging multiple leading pharmaceutical analysts from renowned investment banks and representatives from investment institutions in an insightful sharing and in-depth discussion on the international expansion of innovative drug products and pharmaceutical industry globalization. Participants pointed out that China’s pharmaceutical industry is currently at a strategic inflection point for global expansion. The Intellectual Property (IP) licensing model has preliminarily demonstrated the global competitiveness of Chinese innovation. However, for most domestic pharmaceutical companies, this process remains in the “isolatedbreakthrough” phase. To achieve the transition from a practice of “one-time licensing” to “sustained global engagement,” Chinese pharmaceutical companies must look to multinational pharmaceutical giants as a benchmark — internationalizing their commercialization, manufacturing, and R&D capabilities to build a replicable, scalable, and sustainable global industrial ecosystem.
Achieving this goal is no easy task. While many Chinese pharmaceutical companies have begun exploring emerging markets, they often face challenges such as fragmented distribution channels and difficulty in standardizing operational systems. Only by maintaining conviction and building a fully integrated, internationalized ecosystem across the entire value chain can companies transform one-off licensing revenues into long-term brand equity and control of the value chain, ultimately earning a sustained voice and competitive edge in the global arena.
Though the forum’s spotlight has dimmed, the consensus reached continues to shine like a beacon: Southeast Asia, the Middle East, and other emerging markets are quickly becoming the next major destinations for the global pharmaceutical industry. CMS’s fully localized framework covering “Research, Manufacturing, Commercialization, and Investment” has paved the way for industrial expansion overseas, constructing a bridge to globalization. With an open and win-win attitude, the Group welcomes innovators, regulators, and capital from around the world to work together in bringing more Chinese and global innovative drugs to emerging markets, fostering international growth. CMS sincerely invites partners across all sectors to seize the growth opportunities of the Asia-Pacific region and jointly promote innovation in the pharmaceutical industry across emerging markets, so that more innovative therapies may benefit patients around the world.
Media Contact:
Company: China Medical System Holdings Ltd.
Contact: CMS Investor Relations
Email: ir@cms.net.cn
Website: https://web.cms.net.cn/en/home/
Source: China Medical System Holdings Ltd.