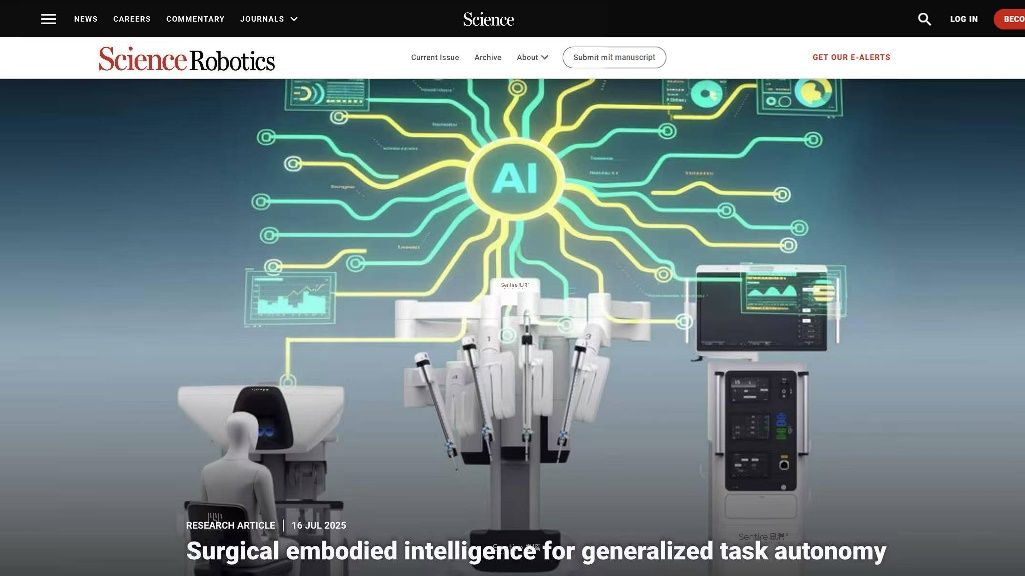
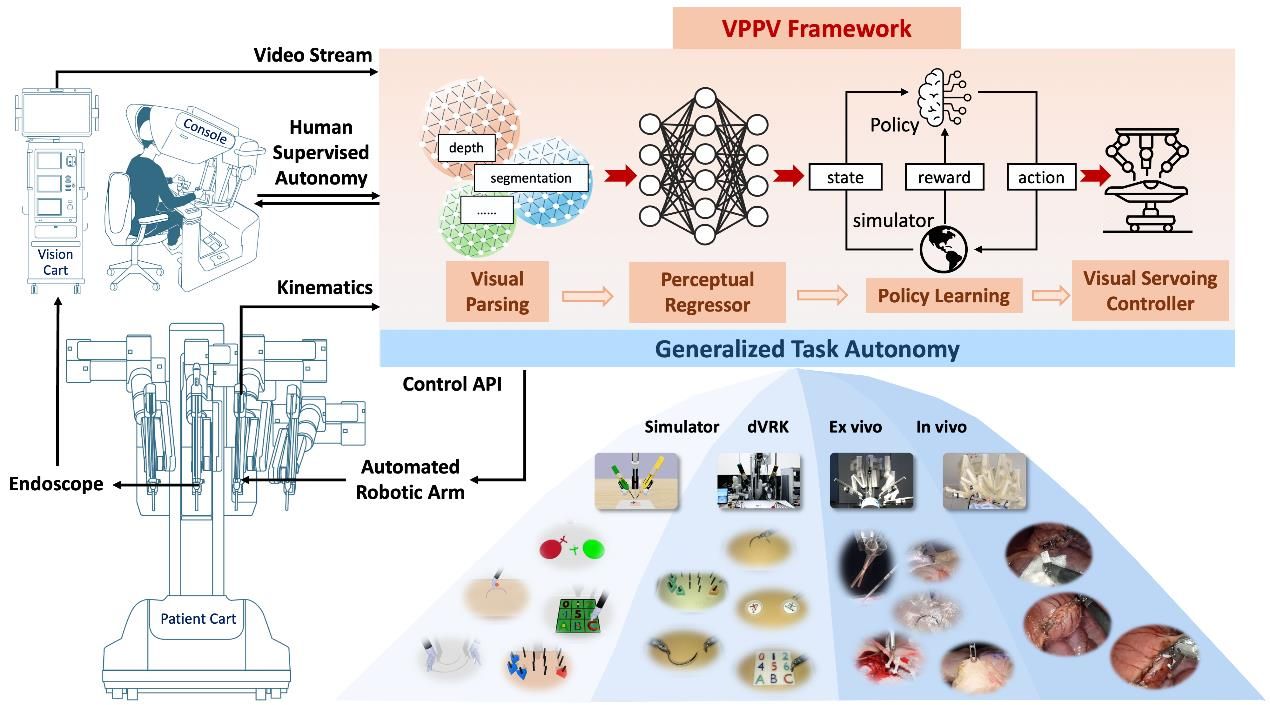
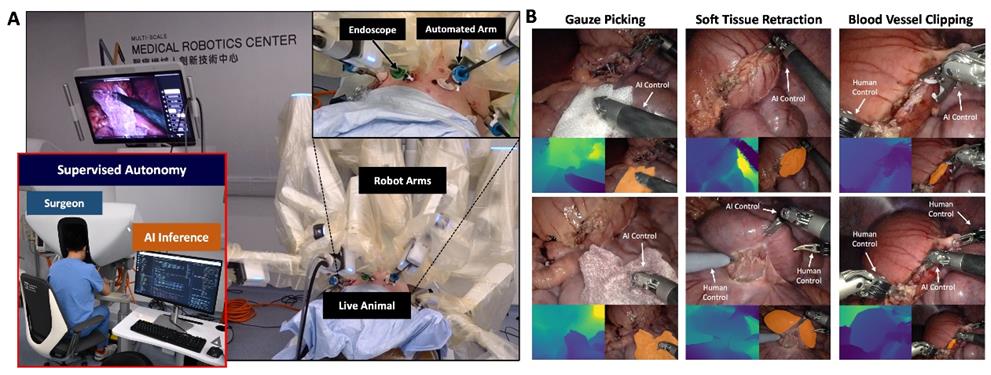
AdipoLABs Co., Ltd. and Red & Blue Co., Ltd. from Korea, together with their Malaysian subsidiary, AdipoLABs Healthcare (M) Sdn Bhd, today officially opened the new AdipoLABs regional office for Asia-Pacific, marking a strategic expansion to strengthen its leadership in non- invasive healthcare and build a robust MedTech ecosystem in Malaysia and across ASEAN.

Picture 1: Top row from left: Adjunct Prof. Dr. Yoo Seung-Mo, CEO, Red & Blue Co. Ltd; Dr. Isaac Joseph, Director of AdipoLABs Healthcare (M) Sdn Bhd; and Tan Sri Dato’ Dr. R. Palan, Pro Chancellor, UoC
Second row From left: Adjunct Prof. Han Sung-ho, President & CEO, AdipoLABs Healthcare Korea; Mr. Moses Balagopal, Director, AdipoLABs Healthcare (M) Sdn Bhd and Professor Dr. David Whitford, Vice Chancellor, UoC
As a research and development-focused medical company, its presence is expected to drive knowledge transfer, foster R&D collaboration, provide industry-standard training, and enhance the country’s preventive healthcare capabilities. To commemorate this milestone, the company also signed two Memoranda of Understanding (MoUs) with the University of Cyberjaya (UoC), formalising a partnership to advance healthcare innovation, clinical research, and talent development.
“By uniting our hyperthermia technology with local expertise and academic rigour, we’ll expedite the adoption of advanced solutions and improve patient outcomes worldwide. We envision this collaboration as a blueprint for international healthcare partnerships,” said Mr. Han Sung-ho, CEO of AdipoLABs Co., Ltd.

Picture 2: Left to right – Adjunct Prof. Dr. Yoo Seung-Mo, CEO, Red & Blue Co. Ltd, Adjunct Prof. Han Sung-ho, President & CEO, AdipoLABs Healthcare Korea, Dr. Isaac Joseph, Director of AdipoLABs Healthcare (M) Sdn Bhd, Professor Dr. David Whitford, Vice-Chancellor, University of Cyberjaya, Mr. Moses Balagopal, Director, AdipoLABs Healthcare (M) Sdn Bhd, Tan Sri Dato’ Dr. R. Palan, Pro-Chancellor, University of Cyberjaya.
A cornerstone of the first MoU is a comprehensive internship programme with UoC’s Physiotherapy programmes under the Faculty of Health, offering six-month placements and potential job opportunities to give students invaluable real-world experience. Students from the Faculty of Health will gain hands-on expertise with AdipoLABs’ Pain Bot device, enhancing their skills in pain detection and treatment, while students from the School of Biomedical Engineering will support AdipoLABs’ technical operations, strengthening after-sales service and device maintenance.
As part of the partnership, AdipoLABs will implement a strategic client engagement initiative, where interns may be placed at client facilities to demonstrate device effectiveness and provide on-site support. This initiative highlights the interns’ value, encourages potential permanent employment, and ensures optimal use of the technology. The collaboration also extends to clinical research, with UoC professors and students conducting studies on various AdipoLABs medical devices, generating evidence-based insights to be published in reputable journals and scientifically validating the technologies.
“We’re thrilled to collaborate with the University of Cyberjaya to cultivate local talent and drive Malaysian healthcare innovation. This partnership offers UoC students invaluable hands-on experience with technologies like Pain Bot. It also expands our clinical research, enhancing pain management solutions for patients across Malaysia and ASEAN. We are dedicated to empowering future healthcare leaders,” said Dr. Isaac R. Joseph, Director of AdipoLABs Healthcare (M) Sdn Bhd.
In a parallel effort to advance physiotherapy education, AdipoLABs Malaysia, Red & Blue Co., Ltd., and UoC signed a second MoU focused on knowledge transfer and medical device donation. Under this agreement, AdipoLABs Malaysia will donate a Pain Bot unit to UoC, giving students access to cutting-edge pain detection and treatment technology and enhancing their knowledge and skills in innovative musculoskeletal pain management methods.
AdipoLABs, a Ministry of Health (MOH)-registered medical-device establishment under the Medical Device Authority(MDA), anchors this collaboration with proven regulatory credibility and a strong commitment to quality and innovation. Their certified standing reinforces UoC’s ambition to accelerate evidence-based research and industry-ready healthcare solutions.
“AdipoLABs’ partnership with the University of Cyberjaya is more than a collaboration, it is a direct investment in the next generation of healthcare professionals. With the introduction of advanced rehabilitative technologies, our students gain access to international-standard tools and real-world innovation that will shape their confidence, competence, and future careers,” said Professor Dr. David Whitford, Vice-Chancellor of the University of Cyberjaya.
Further strengthening this collaboration, Professor Dr. Yoo Seung-mo from South Korea, the inventor of the Pain Bot, will serve as an Adjunct Professor at UoC. Dr. Yoo will provide on-site training and ongoing remote mentorship, ensuring students receive the highest level of expertise in utilising the Pain Bot technology and applying advanced pain detection and treatment techniques.
Together, UoC and AdipoLABs are positioned to drive forward Malaysia’s healthcare innovation agenda with greater confidence and impact.
For more information on University of Cyberjaya or AdipoLABs Healthcare (M) Sdn Bhd, kindly visit: University of Cyberjaya – Premier University in Malaysia or adipolabs-msia.com – AdipoLABs Healthcare Malaysia
ABOUT UNIVERSITY OF CYBERJAYA (UOC)
The University of Cyberjaya is a premier institution located in Malaysia’s first smart city, offering a transformative educational experience on a state-of-the-art, eco-friendly campus. The University was recently ranked in the top 1,000 universities in the world under the QSWorld University Rankings and is also currently ranked top 450 under the QS Asia University Rankings 2026.
The University is renowned for its excellence in medicine, teaching, employability, facilities, and social responsibility. Additionally, it holds a Top 601+ ranking in the Times Higher Education Sustainability Impact Rankings, excelling in Top 200 for Good Health and Well-being (SDG 3).
Founded in 2005 with a focus on medicine and pharmacy, the University has expanded to offer over 50 programmes across diverse fields such as medicine, health sciences, nursing, pharmaceutical sciences, psychology, business, and biomedical engineering. With a global community of over 10,000 students and over 13,000 alumni, the University is shaping future leaders who are making significant contributions to various industries. The University’ impact is reflected in its graduates, who have contributed to innovations in healthcare during the COVID-19 pandemic, participated in international humanitarian missions, and made significant advancements across industries such as business and biomedical engineering.
The University is guided by Chancellor Tan Sri Dato’ Seri Dr. Noor Hisham Abdullah, a prominent figure in global healthcare
for World Health Organization (WHO) initiatives and former Director-General of the Malaysian Ministry of Health. Chairman Professor Tan Sri Dato’ Dr. Mohd Amin Jalaludin, former Vice-Chancellor of the University of Malaya, provides strategic governance as Chairman of the Board of Governors. Under the leadership of Vice Chancellor Professor Dr. David Whitford, the University is strengthening its global outlook and academic excellence. The university can be contacted at +603 8313 7000.
About AdipoLABs Co., Ltd.
AdipoLABs Co., Ltd. is a South Korean medical device company specialising in innovative hyperthermia cancer treatment technology. The company is the manufacturer of the Remission 1℃, a high-frequency hyperthermia device used in hospitals worldwide as an adjunct therapy for cancer treatment. With a commitment to research and development, AdipoLABs strives to improve patient outcomes and contribute to a future free from the burden of disease. AdipoLABs has a global presence, with branches in India, China, and Malaysia.
About AdipoLABs Healthcare (M) Sdn Bhd
AdipoLABs Healthcare (M) Sdn Bhd, established in Malaysia in 2018, is a subsidiary of AdipoLABs Co., Ltd. As the sole distributor of AdipoLABs medical devices in Malaysia and authorised representative for sales, marketing, and after-sales service throughout the ASEAN region, the company is committed to bringing innovative, non-invasive medical devices and innovative solutions to the Southeast Asian market. The company works closely with an international team of researchers, doctors, lab technicians, and engineers to provide cutting-edge healthcare technology to the ASEAN market. Dr. Isaac R. Joseph and Mr. Moses Balagopal are the Directors and Co-founders of AdipoLABs Healthcare (M) Sdn Bhd.
About Red & Blue Co., Ltd.
Red & Blue Co., Ltd. is a South Korean company specialising in the development and distribution of advanced medical devices and healthcare solutions. As a key partner of AdipoLABs Co., Ltd., Red & Blue Co., Ltd. is committed to supporting education and training initiatives that promote the effective use of innovative pain detection and pain treatment techniques. Dr. Yoo works closely with healthcare professionals and academic institutions to advance the field of pain management and improve patient outcomes.
Issued by MNAIR PR Consultancy Sdn Bhd on behalf of University of Cyberjaya and AdipoLABs Healthcare (M) Sdn Bhd Contacts for Media Enquiries:
MNAIR PR Consultancy Sdn Bhd Ameera Hani
Associate Director, Public Relations
+6014 2243296
ameera@mnairpr.com
University of Cyberjaya (UoC) Ehya Nadzeri
Communications
Cyberjaya Education Group Berhad
gcmc@cyberjaya.edu.my