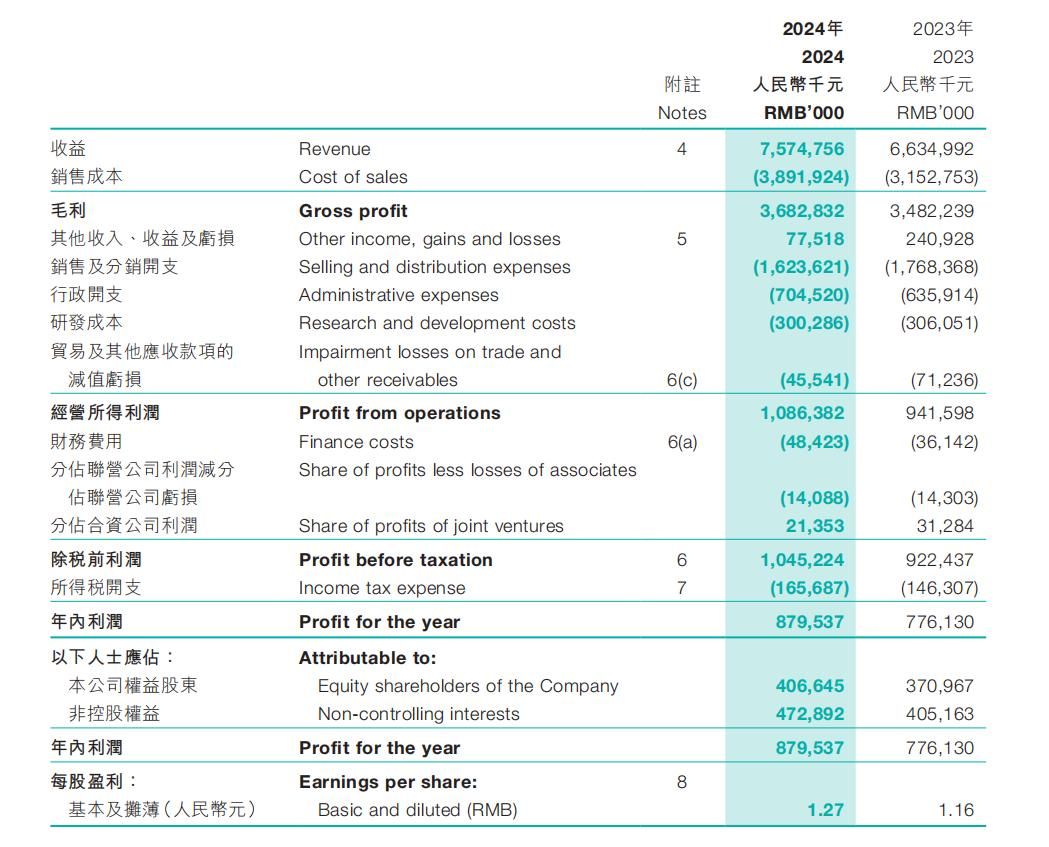
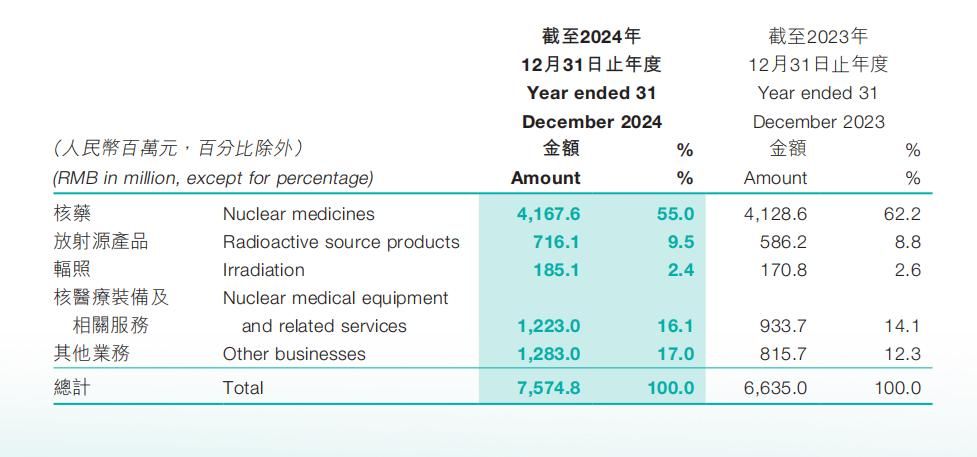
HighTide Therapeutics, Inc. (HKG: 2511), a clinical stage biopharmaceutical company specializing in the development of multifunctional multi-targeted therapies for chronic liver and metabolic diseases, announced today that it will present at the EASL Congress 2025, taking place from May 7-10, 2025 in Amsterdam. The presentations include post-hoc analyses of two Phase 2 clinical studies of berberine ursodeoxycholate (HTD1801), a gut-liver anti-inflammatory metabolic modulator, being developed for treatment of metabolic dysfunction-associated steatohepatitis (MASH) and type 2 diabetes mellitus (T2DM). A third presentation will present preclinical results for rimtoregtide (HTD4010), a peptide derived from the Reg3a protein, in liver failure in mice.
“Effects of Berberine Ursodeoxycholate (HTD1801) in Patients with At-risk MASH and T2DM”
(Presentation SAT-440, Poster Presentation, May 10, 8:30 AM CET)
About the Abstract: Due to the ongoing unmet medical need, clinical development in MASH focuses on patients who are at a higher risk of disease progression and outcomes due to the presence of moderate to advanced fibrosis (defined as at-risk MASH). The purpose of this analysis was to assess the effects of HTD1801 in patients with at-risk MASH and T2DM as defined by baseline MRI cT1 >875 ms. Eighteen weeks of treatment with HTD1801 resulted in substantial improvements in key hepatic and cardiometabolic parameters in patients with at-risk MASH and compared to placebo, twice as many patients achieved a reduction in liver fat content (MRI-PDFF) or fibroinflammation (cT1) that have been associated with improvements in liver histology. These data are particularly insightful as HTD1801 continues to be evaluated in an ongoing paired biopsy study of patients with at-risk MASH and pre-diabetes or diabetes.
“Effects of Berberine Ursodeoxycholate (HTD1801) in Chinese Patients with T2DM and Presumed MASLD”
(Presentation SAT-432, Poster Presentation, May 10, 8:30 AM CET)
About the Abstract: T2DM typically coexists with other metabolic abnormalities such as hyperlipidemia, obesity, and MASH that can exacerbate T2DM and can lead to a worse prognosis with increased risk for mortality and cardiovascular outcomes. In a Phase 2 study in patients with T2DM, HTD1801 achieved the primary endpoint with a significant decrease in HbA1c. Based on the latest diagnostic criteria, it is likely that a substantial subgroup of the study may have had concurrent metabolic dysfunction-associated steatotic liver disease (MASLD). The purpose of this analysis was to evaluate the benefits of HTD1801 in patients with T2DM and MASLD identified by baseline controlled attenuation parameter values >288 dB/M (correlated to 5% liver fat content). HTD1801 treatment demonstrated both dose-dependent improvements in cardiometabolic and hepatic parameters in patients with T2DM and MASLD. These data suggest HTD1801 can comprehensively address metabolic and cardiovascular risk factors beyond glycemic control.
“A Comparison of the Protective Effects of Rimtoregtide (HTD4010) and DUR-928 on Acute Liver Failure in Mice”
(Presentation FRI-141, Poster Presentation, May 9, 8:30 AM CET)
About the Abstract: The purpose of this study was 1) to test the potential protective effects of HTD4010 in an LPS-induced model mimicking acute liver failure in mice and 2) compare these effects to DUR-928, which is currently in late-stage development for the treatment of alcohol-associated hepatitis. In an LPS-induced mouse model mimicking acute liver failure, HTD4010 resulted in significant improvement in survival rates (greater than 2-fold) compared to the model control. These protective effects of HTD4010 were significantly greater than DUR-928. These findings provide evidence that HTD4010 may have a beneficial effect on acute liver conditions including alcohol-associated hepatitis and other acute-inflammatory-related conditions.
About Berberine Ursodeoxycholate
Berberine ursodeoxycholate (HTD1801) is an orally delivered, gut-liver anti-inflammatory metabolic modulator being developed for the treatment of metabolic and digestive diseases. HTD1801, an ionic salt of berberine and ursodeoxycholate, is a new molecular entity with a unique dual mechanism of action: AMP kinase activation and NLRP3 inflammasome inhibition. These two key mechanistic pathways have been associated with improvements in insulin resistance, glucose metabolism, lipid metabolism, and hepatic inflammation, potentially providing a comprehensive treatment platform for the multifaceted nature of complex metabolic diseases. HTD1801 is being developed for multiple indications. HTD1801 met the primary endpoint in two Phase 3 clinical trials in patients with type 2 diabetes mellitus (T2DM), demonstrating a clinically meaningful effect on HbA1c. In both trials, key secondary endpoints were achieved, suggesting multiple advantages of HTD1801 including improvement in cardiometabolic risk indicators. In addition to T2DM, HTD1801 efficacy in treating metabolic dysfunction-associated steatohepatitis (MASH) has been demonstrated in a Phase 2a clinical trial, and a global multicenter Phase 2b trial assessing the histologic benefit of HTD1801 is currently ongoing, with topline results expected in 2025.
About Rimtoregtide
Rimtoregtide (HTD4010) is a clinical-stage compound in development for acute inflammatory-related indications including alcoholic hepatitis (AH). It is a peptide derived from the Reg3a protein with immunomodulatory, anti-inflammatory, and anti-apoptotic effects. HTD4010 has been evaluated in animal models of acute pancreatitis and acute liver failure, where protective effects were observed. A completed Phase 1 clinical trial of HTD4010 in healthy subjects demonstrated a favorable safety profile. AH is caused by chronic heavy alcohol abuse or a sudden, drastic increase in alcohol consumption. It is characterized by severe inflammation and, ultimately, liver failure and death. There is currently no approved treatment for AH and only a few drug candidates are in clinical development. The current standard of care focuses on symptom management, including abstinence, treating inflammation and providing nutrition.
About HighTide Therapeutics
HighTide Therapeutics, Inc. (HKG: 2511) is a globally integrated biopharmaceutical company focusing on the discovery and development of first-in-class multifunctional, multi-targeted therapies with poly-indication potential across metabolic diseases with significant unmet medical needs. HighTide is currently developing several clinical assets and associated global intellectual property rights, and advancing multiple mid-to-late-stage clinical trials including therapies for metabolic dysfunction-associated steatohepatitis (MASH), type 2 diabetes mellitus (T2DM), severe hypertriglyceridemia (SHTG) and primary sclerosing cholangitis (PSC). Berberine ursodeoxycholate (HTD1801), HighTide’s lead drug candidate, received Fast Track designation from the United States Food and Drug Administration for both MASH and PSC and Orphan Drug designation for PSC. HTD1801 has been included in the National Major New Drug Innovation Program under the 13th Five-Year Plan for Major Technology Project in China.
For more information, please visit www.hightidetx.com
Contact: pr@hightidetx.com